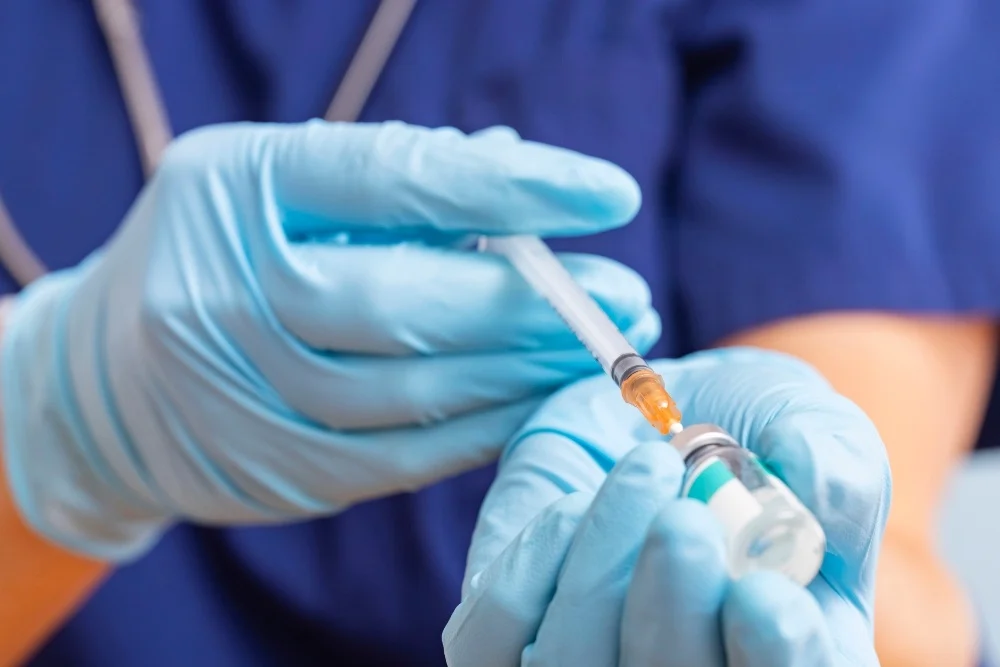
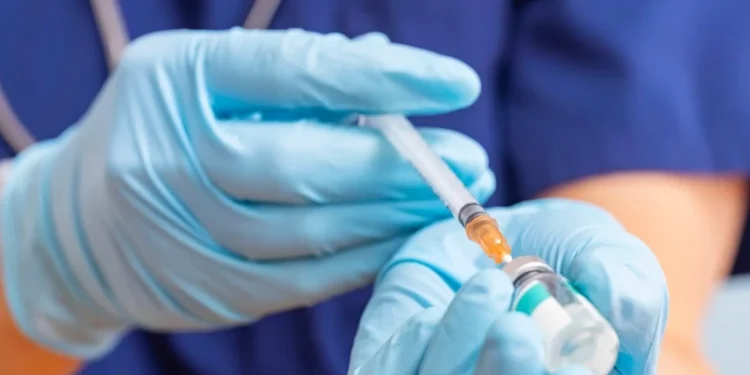
A new modelling study suggests that South Africa could potentially end AIDS as a public health threat by 2032—ten years earlier than currently projected—if a long-acting HIV prevention jab called lenacapavir is rolled out widely across the country. The injection, which only needs to be administered twice a year, has shown 100% effectiveness in preventing HIV among teenage girls and young women in recent trials.
However, to achieve this ambitious target, between two and four million HIV-negative individuals would need to use lenacapavir annually over the next eight years. The jab works by blocking HIV from replicating and invading immune cells, making it highly effective as a pre-exposure prophylaxis (PrEP) method.
Cost and Policy Implications
A study released in March indicates that lenacapavir would be considered cost-effective if priced between R2,238 and R4,304 per person per year. This is 3.5 to 6.8 times more than what the South African health department currently pays for the daily PrEP pill, which costs approximately R629 per person annually. The health department is awaiting further pricing information and regulatory approvals before making procurement decisions.
The World Health Organization is expected to issue a recommendation once regulatory approvals in the United States are finalised. South Africa’s own medicines authority is also reviewing the application as part of a joint international regulatory effort.
Setback in Global Funding
The promising outlook for lenacapavir comes at a difficult time for international HIV prevention efforts. The United States’ President’s Emergency Plan for AIDS Relief (PEPFAR), which has funded 91% of HIV prevention globally, has not been reauthorised by Congress. As a result, funding for many prevention programmes, including support for PrEP, has been frozen or cut altogether. U.S. foreign aid policy now limits the use of these funds for HIV prevention only to pregnant or breastfeeding women.
Advocates warn this shift risks reversing hard-won gains. Vulnerable populations—such as young women, LGBTQ+ individuals, sex workers, and people who inject drugs—are now at higher risk of losing access to life-saving prevention tools.
Despite this, the Global Fund to Fight AIDS, Tuberculosis and Malaria has pledged to honour its agreement with Gilead Sciences to procure lenacapavir for distribution in high-incidence, low-income countries, including South Africa. The deal covers enough doses for two million people over the next three years, pending availability of generics after 2027.
A Game-Changer for Women and Girls
One of the most compelling aspects of lenacapavir is its appeal to young women, who make up 40% of new HIV infections in South Africa but only 8% of the population. In 2023 alone, 122 girls and young women were infected with HIV daily. Studies show they overwhelmingly prefer the twice-yearly injection over a daily pill, citing its discretion and ease of use.
Trials have demonstrated that the jab works just as well in adolescents aged 16–17 as it does in young adults. The injection has also proven effective for gay and bisexual men and transgender people, further highlighting its broad potential.
Could It Really End AIDS?
The modelling analysis shows that if lenacapavir reaches its target audience consistently, it could cut new infections in South Africa by up to 41% over the next two decades. This would bring HIV incidence below the 0.1% threshold, a key benchmark in declaring AIDS no longer a public health crisis.
To reach this goal, South Africa will need a strong political and financial commitment—not just from international donors, but from the government itself. Experts stress the urgency of local investment, especially now that U.S. support is uncertain.
South Africa already leads the world in the use of oral PrEP, with 1.78 million people having used the daily pill. However, adherence remains a challenge, particularly for young women. Many struggle to take the pill daily or remain on it for extended periods—an issue lenacapavir’s long-acting formula could solve.
Looking Ahead
South Africa’s National Essential Medicines List Committee is currently reviewing the cost-effectiveness of lenacapavir. While the drug is not yet registered locally, it is undergoing an accelerated joint review by the European Medicines Agency, WHO, and South Africa’s own regulator. Once approved, it will take about three additional months for local clearance.
In the meantime, health advocates are urging South Africa to prepare for implementation, ensuring systems are in place to reach the millions who could benefit. With the right planning and investment, lenacapavir could become the turning point in South Africa’s decades-long fight against HIV.
As HIV prevention advocates argue, the window of opportunity is here—but it may not stay open for long.